Submitted by Helen C. Scott on Fri, 12/08/2016 - 12:00
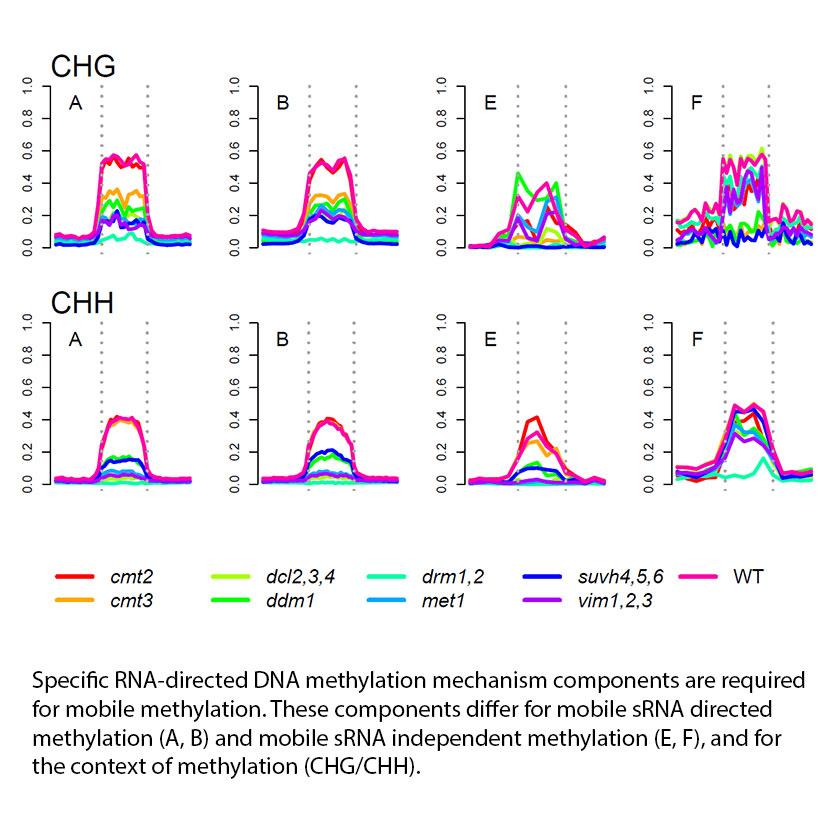
Small (s)RNAs of 21 to 24 nucleotides are associated with RNA silencing and methylation of DNA cytosine residues. All sizes can move from cell-to-cell and long distance in plants, directing RNA silencing in destination cells. Twenty-four nucleotide sRNAs are the predominant long-distance mobile species. Thousands move from shoot to root, where they target methylation of transposable elements both directly and indirectly. In this follow up to our earlier work establishing the mobility of small RNAs and their ability to regulate methylation at thousands of locations, we examine the characterisation of distinct classes of siRNA/methylome interactions. Comparisons to methylation in RNA silencing knock-out mutants demonstrate that methylation loci regulated by a mobile sRNA signal depend upon different elements of the establishment and maintenance phases of RdDM than do those loci whose methylation is not regulated by the mobile signal. We further demonstrate that locus classes differ in the histone marks with which they are associated. Methylation directly associated with mobile sRNAs, and mobile sRNAs generally, associate with H3K27me3, a mark found typically in repressed regions of the genome, whilst loci methylated through indirect effects of mobile sRNAs associate with H3K36me2 and H3K36me3, both of which are found typically in active regions of the genome. In contrast, the global sRNA population is associated with the H3K9me2 mark. The association of mobile sRNA loci with H3K27me3 may account for the reduction in expression across genes overlapping mobile sRNA loci, as this occurs even when the mobile sRNAs are not present, as in the dcl234/dcl234 graft.
We have thus shown that the pathways by which mobile sRNA signals regulate methylation are fundamentally distinct to those independent of a mobile signal. Future investigations might focus on characterising functional differences between direct and indirect loci, as well as how the indirect loci are regulated. The manner of mobile sRNA transport and their influence upon phenotypes, molecular or morphological, also remain to be elucidated.
- Hardcastle, TJ, Lewsey MG. Mobile small RNAs and their role in regulating cytosine methylation of DNA. RNA Biology (2016) (http://dx.doi.org/10.1080/15476286.2016.1218591)
